Introducing: The Biotech Tea
What's brewing in biotech this week: through the lens of a biotech scientist!
I’m very excited to introduce The Biotech Tea as a regular installment here! Each week, I’ll share a quick rundown of biotech breakthroughs, policy shifts, and the science shaping future medicines.
Most of you probably know—but maybe some of you don’t—that I’ve worked as an industry scientist since 2019, after finishing my PhD. I’m part of an analytical development team at a large biotech company based in Boston. I love the work I do there.
Over time I’ve realized that the biotech industry can feel like a bit of a black box to people outside of it. My hope is that The Biotech Tea can help open that box—keeping those of you interested in biotech up to date on what’s happening, through the lens of a scientist who gets to see the inside.
A quick description of the newsletter - I’ve decided on 5 key pillars that will be covered each week and 1 bonus content for paid subscribers.
Story I’m watching: what caught my attention this week.
From the Bench: an exciting science update.
Bio[Tech]: an advancement worth noting.
The Rulebook: an important standard or regulatory development.
The Human Side: a patient or scientist perspective worth highlighting.
and bonus for paid…
Biotech Term of the Week: helping you speak biotech.
As always, I appreciate you being here and hope that you find this perspective on science and public health updates to be helpful.
So without further ado…
Story I’m Watching
What caught my attention this week.
Have you heard the names Şahin and Türeci before? If you follow any science communicators since the pandemic you likely have because they are some of the key scientists behind mRNA vaccine technology. They are co-founders of BioNTech, most well known for COMIRNATY, the COVID-19 mRNA vaccine developed with Pfizer. From their recent announcement it seems like these two remarkable innovators are looking to step away from late-stage development and commercialization (which is the usual bread and butter of larger companies) to go back to early-stage research (more a start-up or small-company focus).
“Özlem and I are ready to become pioneers once again.” - Şahin
Excitingly, they plan to start a new company focused on next-generation mRNA innovations. I will be watching for it to get started and look forward to seeing what comes out of it!
From the Bench
An exciting science update.
A paper published this week in Nature demonstrated a new method (dubbed INSTALL) for safely incorporating large sequences of DNA into genomes.
What’s new about it? Circular single stranded DNA (cssDNA) as opposed to double-stranded DNA (dsDNA).
And why does that matter? dsDNA detected in the cytoplasm of a cell triggers the innate immune system. cssDNA, on the other hand, is able to evade the cellular immune triggers.
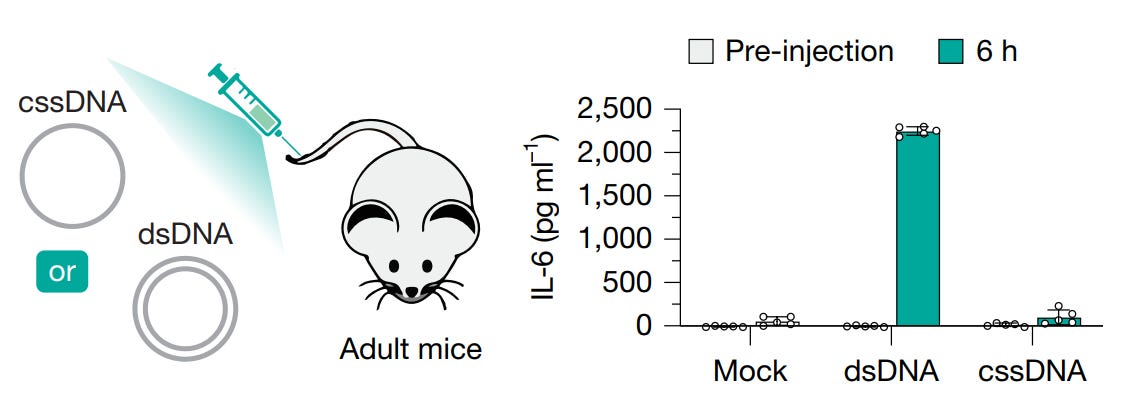
The team behind this work was inspired by certain bacteria and viruses that are able to naturally insert cssDNA into double stranded genomes and they developed a method for doing so in human cells!
“This is beautiful work that addresses in an elegant way one of the big challenges with controlled DNA insertion into the genome,” Kiran Musunuru, M.D., Ph.D., co-founder of Verve Therapeutics and a scientist at the Children’s Hospital of Philadelphia and the University of Pennsylvania.
Bio[Tech]
An advancement worth noting.
Meet Spectris, a sensory stimulus device developed by Cognito, worn on the head for the treatment of Alzheimer’s Disease.
For some context: the only two treatments on the market for Alzheimer’s are from Eli Lilly (Kisunla) and Eisai/Biogen (Leqembi) - both targeting amyloid-β. Importantly, these are both pretty new to the market (within about 3 years) and nothing existed for treatment before.
So what about Spectris? Data from a 673 person randomized control trial is nearing completion and results should be in later this year. The company says they’d be looking to file through the de novo pathway hopefully later this year with it available to patients, assuming it is approved, by mid-2027.
I will be watching for the data and will report back when I hear!
The Rulebook
Am important standard or regulatory development.
Earlier this week the FDA announced the launch of a new adverse event look-up tool - AEMS, short for Adverse Event Monitoring System. The idea is to have a single streamlined dashboard for drugs, biologics, vaccines, cosmetics, and animal food. Reporting will need to transition to AEMS in the coming months and historical AE data will be migrated over.
What are my thoughts? If structured well, a centralized location for AEs would make data analytics easier. I think my concern lies with the both:
The data quality of the original databases
The quality of the data migration
I have been involved in some massive documentation migration initiatives at work (moving from an old document system to a new) and the effort was no small task. Historical data is often missing important metadata that makes it hard to properly categorize in new systems. We already know (from ample pandemic exposure) that VAERS reports can be entered by anyone, and the idea of cleanly migrating that data over sounds impossible.
The following paragraph from EndPoints News further suggests that data coming from VAERS will lead to data bias.
“The CDC began to include secondary reports in the database last year — instances in which a VAERS report for the same person, vaccine and dose came from a second independent source. The agency warned that these would show up as additional reports in the database, which could be misleading in a raw tally.”
I will be watching for more information!
The Human Side
A patient or scientist perspective worth highlighting.
I was thrilled to see this opinion piece by a dear friend and fellow scientist, Liz (Dr. Elisabeth Marnik), last month. Titled ‘I Grew Up Unvaccinated. Now I’m an Immunologist.’, Liz eloquently describes the perspective of her mother when she chose not to vaccinate her children. It’s a beautiful story meant to teach us about how important it is to communicate science with empathy.
“When I hear stories about children getting sick from measles, I feel angry. But not at the parents. I’m frustrated at those knowingly spreading false and misleading claims, and profiting from it. Shaming and dismissing parents who are confused and trying their best won’t change minds. Instead, it can drive people deeper into communities that validate their beliefs and create echo chambers filled with inaccuracy. We have to meet parents’ concerns with empathy. That’s where real change begins.” - Dr. Elisabeth Marnik
Biotech Term of the Week
Helping you speak biotech.
Paid Subscriber Bonus Content (for the first few installments it will be free to preview!)
RESIDUAL TESTING
non biotech synonym: looking for contaminants
When a batch of drug is made, it must go through a panel of tests to assess quality and consistency. This panel varies by drug depending on what it is an how it is made. “Residuals” are things that were introduced during the production process that aren’t intended in the final product but even after careful steps to remove, may exist in small amounts.
“process-related impurities that remain from manufacturing but should be removed or controlled before release”
Residuals can be things like solvents used during synthesis (e.g. ethanol, dimethylformamide) or reagents used during purification (e.g. enzymes, detergents). For biologics residuals expand into things that can come from the production cell line (e.g. host-cell protein, host-cell nucleic acid, endotoxins).
Regulatory agencies set clear limits on each residual based on known toxicity of the residual, dose of the drug in the patient, and the route of administration (how it will be delivered).
What’s my experience with residual testing?
I have helped oversee the development of residual DNA methods and have seen the development of residual protein methods. The goal for any method, but particularly for residual methods, is to develop something with incredible accuracy and precision with a very low threshold so we are able to pick up even the smallest amounts.
Importantly, not all DNA or protein is equal. If there is concern that a particular sequence or peptide may be present that has some known toxicity or mechanism of action, development groups (ahem analytical development) will design methods to look for them so risk assessments can be done early. Communication with regulatory agencies is fluid to set the right limits for residuals.
Here’s an article on residual testing if you’d like to read more.




